Acids, Bases, and Salts You should be able to Understand the acid-base theories of Arrhenius, Brønsted-Lowry, and Lewis. Identify strong acids and. - ppt download

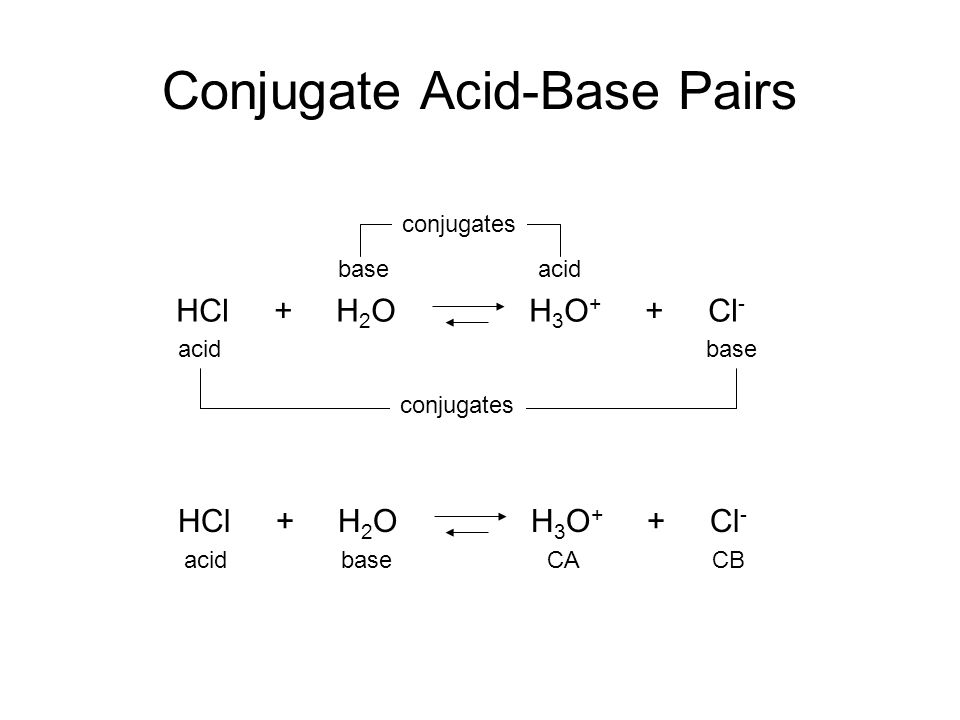
SOLVED: Fill out the conjugate acids/bases in the following table: Conjugate Acid Base Acid: HF Conjugate base: F- Base: SO4 Conjugate acid: HSO4- Base: Cl Conjugate acid: HCl Acid: HCIO Conjugate base: