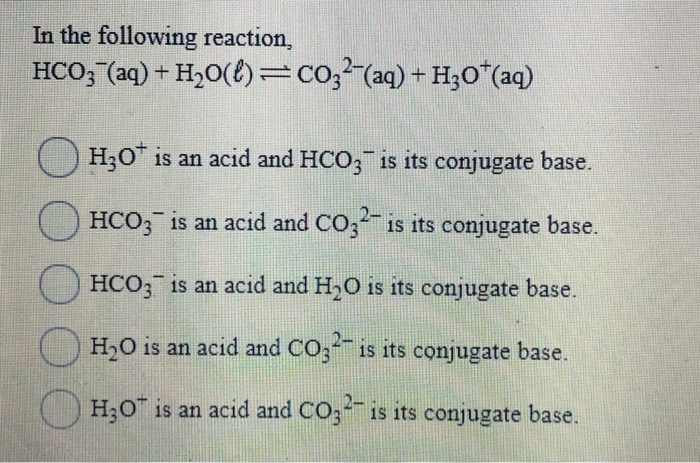Réaction Acide-base. Réaction Chimique Neutralisant Les Propriétés Acides Et Basiques, Produisant Un Sel Et De L'eau. Utilisé Pour Déterminer Le Ph. Bronsted - Théorie De Lowry. Molécules De Hcl, Naoh, H2o Et

SOLVED: QUESTION 11 In the following reaction: HCO3" (aq) + H2O (aq) -> H2CO3 (aq) + OH" (aq) 0 HCO3" is an acid and is H2CO3 its conjugate base. 0 H20 is

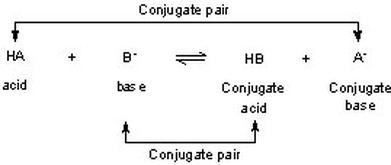
SOLVED: B. Identify the conjugate acid-base pairs in the following reactions: H2PO4- + OH- → HPO4-2 + H2O HBr + H2O → H3O+ + Br- CO3-2 + H2O → HCO3- + OH-